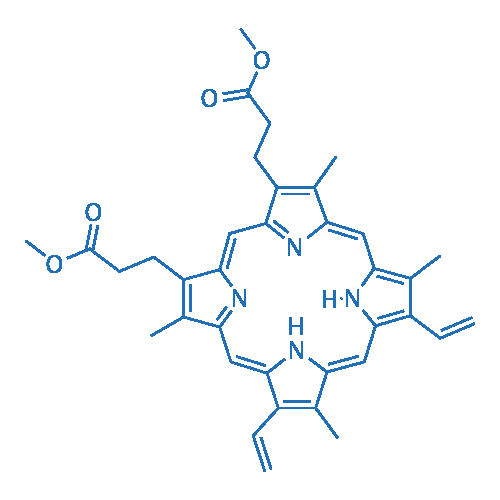
Protoporphyrin IX: the Good, the Bad, and the Ugly | Journal of Pharmacology and Experimental Therapeutics
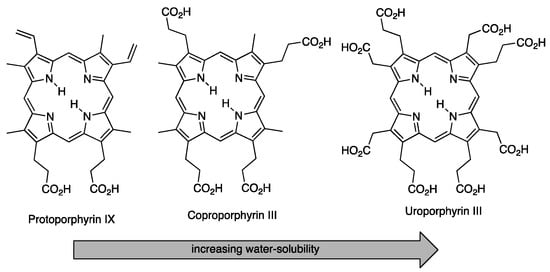
Molecules | Free Full-Text | Modifications of Porphyrins and Hydroporphyrins for Their Solubilization in Aqueous Media | HTML

Protoporphyrin IX–gold nanoparticle conjugates as an efficient photosensitizer in cervical cancer therapy - ScienceDirect
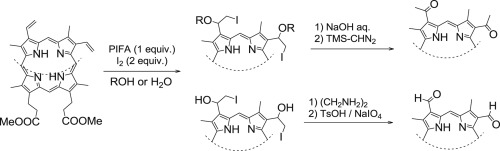
Facile iodination of the vinyl groups in protoporphyrin IX dimethyl ester and subsequent transformation of the iodinated moieties - Tetrahedron - X-MOL

US20080242857A1 - Process For Preparing Porphyrin Derivatives, Such As Protoporphyrin (IX) And Synthesis Intermediates - Google Patents
![3-[18-(2-Carboxyethyl)-7,12-bis(ethenyl)-3,8,13,17-tetramethylporphyrin-21,23-diid-2-yl]propanoic acid;palladium(2+) | C34H32N4O4Pd - PubChem 3-[18-(2-Carboxyethyl)-7,12-bis(ethenyl)-3,8,13,17-tetramethylporphyrin-21,23-diid-2-yl]propanoic acid;palladium(2+) | C34H32N4O4Pd - PubChem](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=472085&t=l)
3-[18-(2-Carboxyethyl)-7,12-bis(ethenyl)-3,8,13,17-tetramethylporphyrin-21,23-diid-2-yl]propanoic acid;palladium(2+) | C34H32N4O4Pd - PubChem
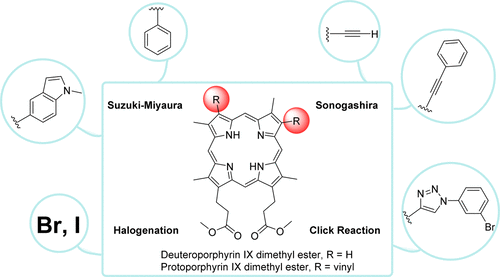
Functionalization of Deutero- and Protoporphyrin IX Dimethyl Esters via Palladium-Catalyzed Coupling Reactions - J. Org. Chem. - X-MOL
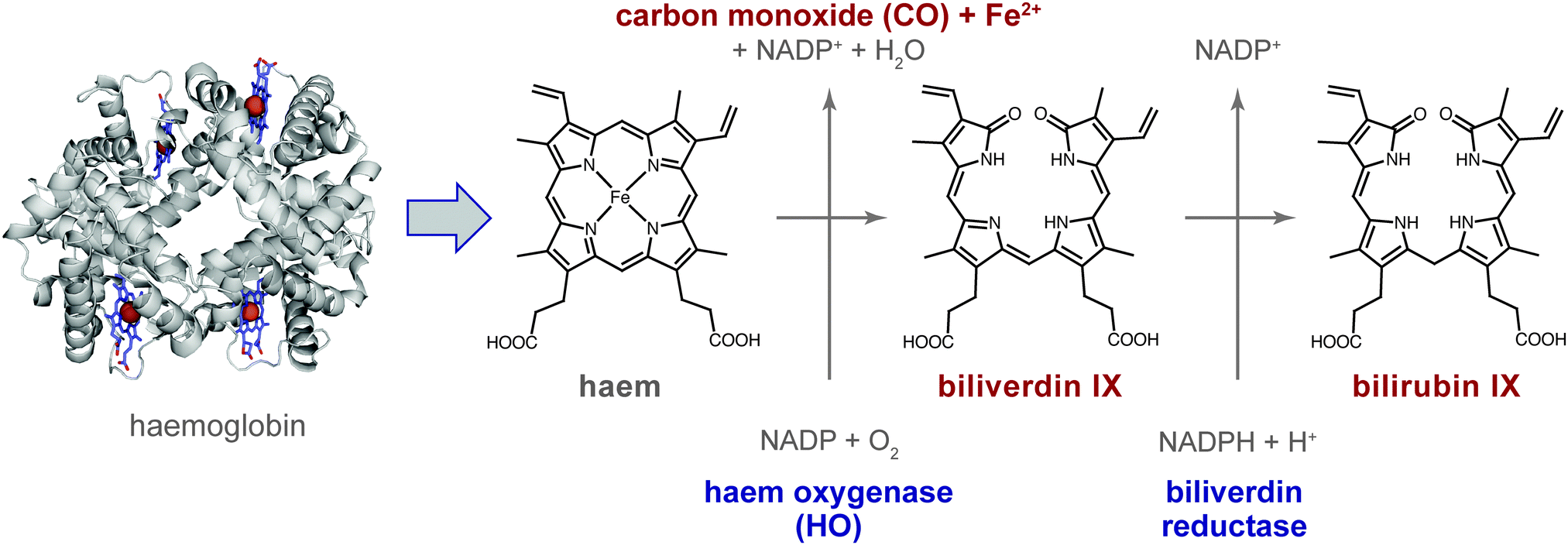
Carbon monoxide – physiology, detection and controlled release - Chemical Communications (RSC Publishing) DOI:10.1039/C3CC49196J

US20080242857A1 - Process For Preparing Porphyrin Derivatives, Such As Protoporphyrin (IX) And Synthesis Intermediates - Google Patents

US20080242857A1 - Process For Preparing Porphyrin Derivatives, Such As Protoporphyrin (IX) And Synthesis Intermediates - Google Patents

Protoporphyrin IX–gold nanoparticle conjugates as an efficient photosensitizer in cervical cancer therapy - ScienceDirect

WO2019190787A1 - Method for stabilizing hemoglobin and reagents for performing the same - Google Patents

PDF) Functionalization of Deutero- and Protoporphyrin IX Dimethyl Esters via Palladium-Catalyzed Coupling Reactions